|
How to Families Work
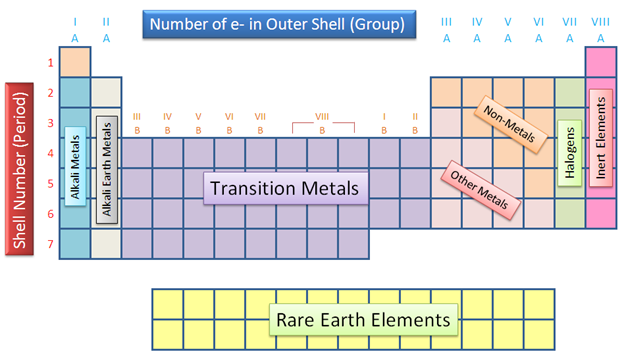
The Periodic Table is separated into 8 families: Alkali Metals, Alkali Earth Metals, Transition Metals, Other Metals, Non-metals, Halogens, Rare Earth Metals, and Nobles Gases. From left to right the elements are ordered from most active to least active elements. Click link to go to Families |
|
Rows and Columns
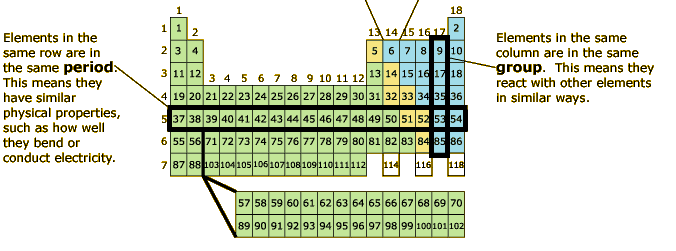
Elements in the same row share similar properties like how well they conduct heat and electricity. The numbers over the columns state how many valence electrons are on the outer most shell of each element, except for transition metals. The left most columns have the least number of valence electrons and tend to give away their electrons when combining with another element. On the other hand, the columns more to the right tend to have more valence electrons and take electrons when merging with other elements. |
|
How to Read Each Element
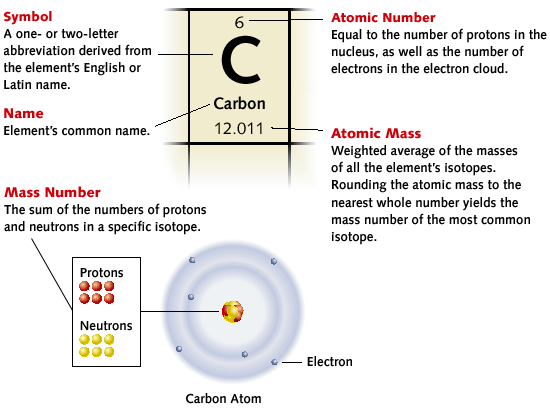
Each element is unique. To identify these elements, each box on the table has an atomic number, element symbol, element name, and atomic mass. The atomic number, located on the top of the box, is the number of protons the element has or the number of electrons if the atom is neutral. The huge one or two-letter abbreviations, located in the center of the box is the symbol of the element, either deriving from the elements English or Latin name. Under the element symbol, rests the name of the element, followed by the atomic mass. The atomic mass, is the average weight of all the elements isotopes, and when subtracted from the atomic number, give a person the number of neutrons in the element. |
By: Amber Li