|
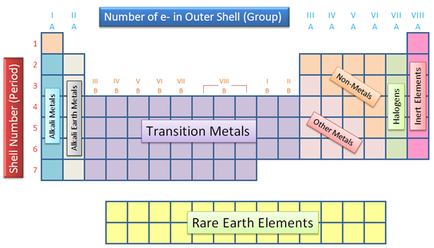
Alkali Metals Highly reactive metals found in group one. These atoms only have one electron in their outermost shell. Alkaline Earth Metals are ready to lose that one electron while bonding with other elements. Same with every metal, the alkali metals are malleable, ductile, and are good conductors of heat and electricity. Alkaline Earth Metals These earth elements are metallic elements found in family number two. They are very reactive because they have two plus oxidation numbers. Transition Metals Thirty-eight elements in groups three through twelve in the periodic table are called transition metals. Same with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat. Their valence electrons are present in more than one shell. This is why they often exhibit several common oxidation states. Other Metals The "other metals" elements are located in groups 13, 14, and 15. While these elements are ductile and malleable, they are not the same as the transition elements. These elements, unlike the transition elements, do not exhibit variable oxidation states, and their valence electrons are only present in their outer shell. All of these elements are solid, have a relatively high density, and are opaque. Metalloids Metalloids are the elements found between the black zig zag line going up and down the periodic table. Metalloids have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors. While other elements are not. Metalloids Metalloids are the elements found between the black zig zag line going up and down the periodic table. Metalloids have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors. While other elements are not. |
Halogens
The halogens are five non-metallic elements found in group 17 of the periodic table. All halogens have 7 electrons in their outer shells. Noble Gases The noble gases are found in group 18 of the periodic table. These elements have an oxidation number of 0. This prevents them from forming compounds readily. All noble gases have 8 electrons in their outer shell, making them stable. Rare Earth Metals The 30 rare earth elements are composed of the lanthanide and actinide series. One element of the lanthanide series and most of the elements in the actinide series are synthetic, that is, human-made. All of the rare earth metals are found in group 3 of the periodic table, and the 6th and 7th periods. |
By: Jack Setran